 
One such discovery was that of atomic masses. The modern periodic table didn’t spring fully formed from the genius of Mendeleev it was shaped by key discoveries about the elements. So how did Russian chemist Dmitri Ivanovich Mendeleev and the other creators of the periodic table (arguably six of them) bring order to the elements? How did they create a tool that would ultimately house 118 elements when they knew only 62 of them? And why does Mendeleev get all the credit? Mass + reactions = periodic table It even predates knowledge of the noble gases. 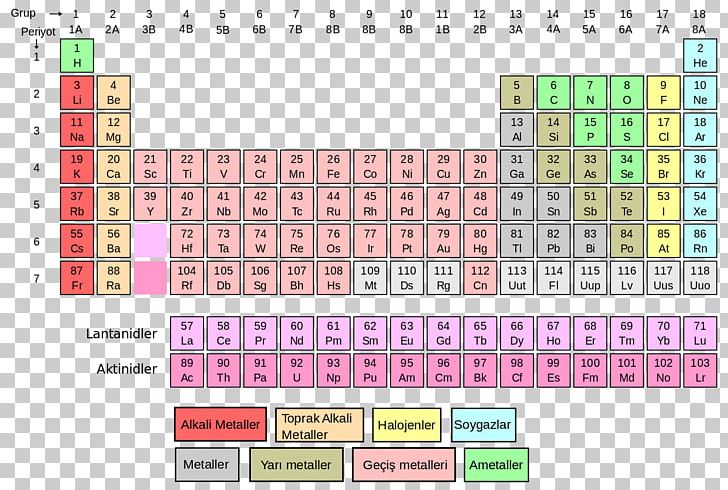 
However, the periodic table predates knowledge of atomic numbers and subatomic particles (yes, including electrons). If you know about atomic numbers and electron shells, recreating the periodic table is simple. You would order them by increasing atomic number and create a new row when you hit a noble gas. If I gave you all of the elements on cards and told you to recreate the periodic table, you probably wouldn’t have much trouble. You can compare electronegativity, ionization energy, atomic radius, chemical reactivity, and more. You can determine the electron configuration of any atom, simply from its place. Interested in learning more about the possibilities of hydrogen education? Discover how the Horizon Hydrogen Grand Prix (H2GP) program can benefit students in a classroom near you.The periodic table is an elegant demonstration of properties of elements. The result is for every 1 unit of hydrogen produced, 8 units of oxygen are created. Why does a hydrogen generator (electrolyzer) actually produce 8 times more oxygen than hydrogen? The molar mass of 2 molecules of water (2 H 20) is 18, and once the hydrogen generator splits the water into hydrogen and oxygen, the hydrogen (having a low atomic mass) only has a mass of 2 (H 2), while the oxygen has a mass of 16 (O 2). Students can understand, for instance, the process of hydrogen generation (electrolysis) with greater insight. Knowing the molar mass of hydrogen will open up a world of opportunity in terms of hydrogen education. Because of this apparent inertness, the H 2 molecule can be considered a very stable molecule at room temperature. Molecular hydrogen (H 2) can also react with many elements and compounds, but at room temperature, the reaction takes place at such a slow rate it's almost negligible. This is how hydrogen molecules – H 2 – are formed. Because of this reactivity, atoms of atomic hydrogen (H) combine with each other to form molecules of two atoms (H 2). This means that overall hydrogen is an extremely ‘light’ element. The more protons an atom has in its nucleus, the higher the atomic number, and the element also becomes heavier. As hydrogen gas is formed of two hydrogen (H) molecules bound together (H 2) – the molar mass can be calculated as 1.00784 x 2 = 2.01568 grams.Īs hydrogen consists of only one proton, its atomic number is 1. It can be calculated by multiplying the atomic weight of hydrogen (1.00784 atomic mass units) by 2. The molar mass of hydrogen is 2.01568 g/mole. This means you can simply multiply this by 4 moles, and you’ll find out how much carbon you’ll need to weigh. You already know carbon has a molar mass of 12.01 g/mol. Take this example: say you need 4 moles of pure carbon for an experiment. 
This means whether you’re trying to figure out how much carbon, titanium, or cobalt you need for an experiment – understanding the molar mass is essential. Molar Mass is defined as the ratio between the mass and the amount of substance (measured in moles) of any sample of a compound. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
